The lens in your eye can change its optical power: when you focus somewhere on the vision, this is what happens. Payback for the «moving parts of the mechanism» — wear with age. At about 40-45 years old, problems usually begin with age-related visual impairment; in 60-70 years, you need up to three pairs of different points. At the same time, the clarity of view is reduced due to the process of sclerosis and lens opacity — it is worth thinking about replacing the lens, until it becomes dim and becomes completely rigid.
To date, the technology of replacing the lens is standardized and well developed. Potential risks are generally somewhat higher than with laser vision correction, but at present this type of surgery is performed even on transparent lenses with refractive purpose (when the patient does not like to wear glasses with thick glasses), that is, it is quite a “household” task for ophthalmology.
What is an artificial lens?
This is a piece of acrylic (or silicone, or polymethyl methacrylate (PMMA), or even some polymer), implanted into the cavity of the eye. The softness of the implant depends on the material from which it is made. Conventionally, models are divided into rigid, that is, incapable of “folding” and therefore implanted through wide cuts in the “original” form, and “soft, elastic” — capable of rolling into a “roll”. For example, a “hard” non-deformed PMMA lens can be placed inside the eye through an incision of 6.2 — 6.5 millimeters. In this case, the cut should correspond to the diameter of the optical zone — and the optical zone less than 6 millimeters gives a large optical distortion.
Soft lenses began to implant historically through incisions of 3.75 mm, folding them in half with tweezers. Then they moved to smaller cuts, more similar to a 2.75 — 2.5 mm puncture, because they were made with a single instrument — a microkeratome, creating a stepped self-sealing incision. Thus began the era of seamless cataract microsurgery.
In fact, all the time in cataract surgery, the development of its removal technology goes side by side with the development of materials for intraocular lenses. If the lens is removed after 6 mm (as it was with extracapsular cataract extraction — EEC), then you can be satisfied with a rigid intraocular lens with 6 mm optics. With the advent of phacoemulsification through 2.75 mm, the need arose for such lenses that fold to a size of 2.5 mm. Previously, there was simply no need: the diameter of the cut is a limiting factor. And when we switched to microfaco emulsification through 2.0 — 1.8 mm, the lenses became more flexible and the systems for their implantation became even more jeweler. By the way, when I say we, I mean clinics where equipment is being modernized according to current trends. But still enough places
Today we remove a cataract of any density through a puncture of 1.7 mm — 1.8 mm (which requires quite expensive consumables), many clinics in Russia work with a puncture length of 2.2 — 2.0 mm (this is also a good indicator). For these sizes, special lenses are needed that can be implanted through a puncture of this magnitude — these are more modern types of lenses. It's a shame if for implantation it is necessary to expand the incision for models of older generations of IOL. The experience and skills of the surgeon are also decisive — the lens itself does not sink into the patient's eye.
So, this is what happens before and during surgery
1. The patient comes to the clinic for diagnosis. If it turns out after it that a lens has to be replaced, the patient has time to think — usually, with the exception of injuries and serious pathologies, the operation suffers weeks. We need quite a serious comprehensive diagnosis for the correct choice of the model and the calculation of the lens. In addition to a general study of optics of the eye, control of intraocular pressure, examination of the fundus with an enlarged pupil, this complex includes the study of visual fields, counting endothelial cells of the cornea, optical or ultrasound biometry, ultrasound scanning of the vitreous body and retina, often optical coherent tomography and corneal keratotopography. This whole set of studies allows you to make the right choice of intraocular lens.
2. Much attention is paid to the discussion with the patient about the distance at which he needs to have vision without correction with glasses. We remember that a new lens with monofocal optics is calculated on a specific focus (as a rule, it is a distance), and in this case you will need glasses for reading. If a patient has an active lifestyle and a great desire not to use glasses either for distance or near, then a multifocal lens is needed. If there is astigmatism, then to compensate for it, the most correct choice of IOL is the astigmatic (toric) lens. If there is both astigmatism and the desire to get rid of glasses, then the choice is the most difficult — the multifocal astigmatic lens.
Another surgeon always takes into account the state of the retina, the safety of the lens ligaments, the state of the cornea — in general, the expert should be a competent surgeon, not a neighbor or a friend
3. The day before the operation, a preparatory complex of laboratory and instrumental examinations of general therapeutic nature is carried out.
On the day of surgery, the patient leads a normal life — he eats breakfast, takes medications prescribed by other doctors and arrives at the clinic at the appointed time.
He spends several hours in the clinic; most of this time is spent preparing for the operation — pupil dilation, pressure control, and other manipulations.
The duration of the operation with a standard cataract is 10-15 minutes. It is performed under local anesthesia, often only drops are sufficient. During the operation, the patient can talk himself and hears the voice of the surgeon, sees the light in front of the eye and feels watering. This is a painless procedure. In some cases, of course, you can perform the operation under general anesthesia, but for this you need to have medical indications or a patient’s desire. And if there is no evidence, then I do not recommend doing so — 10 minutes of anxiety are not worth it. The main manipulations will not be visible — tools rarely get into focus. Even the most anxious patients endure the surgery very calmly.
4. After surgery after an hour and a half, you can go home on your own, but we recommend doing this with the accompaniment, since on the way home the operated eye remains covered with a bandage. With a drop of drops, which must be dripped into the operated eye every 3 hours — it turns out 5 times a day.
5. The next morning, you need to come to the first postoperative examination, subsequent visits are scheduled once a week for a month. Selection of glasses is also recommended not earlier than 3-4 weeks, since after any operation the optics of the eye is restored gradually.
How is the operation of the phacoemulsification of the lens
The idea of ultrasonic phacoemulsification of the lens appeared in the American ophthalmologist Charles Kelman, when he was in the dentist’s chair, where ultrasound was used to clean the teeth. He thought — why not crush a dense lens with energy, say ultrasound, through a small hole and remove through it the same? It took a lot of perseverance and great luck to translate the idea into reality. It was in the 60s of the last century. In 1967, the publication “Phacoemulsification and aspiration.” Appeared in the “American Journal of Ophthalmology”. A new technique of cataract removal. A preliminary report on a new technique for removing cataracts using low-frequency ultrasound in an experiment in the eyes of animals. Kelman together with the company Cavitron (CavitronKelman irrigation and aspiration phacoemulsification system — a prototype of modern phacomachines) developed and patented in 1971 the first model of phacoemulsifier. It consisted of an electronic ultrasonic energy generator unit (frequency 40 kHz) and a nickel piezoelectric transducer with replaceable needles with channels for irrigation and aspiration. So, by the way, this year, on May 8, the technology of ultrasonic phacoemulsification of the lens turned 50 years old.
Modern machines, like the original Kelman model, consist of an electromagnetic generator with a handle with a titanium needle connected to it, which oscillates in the longitudinal direction at ultrasonic frequencies. The lens collapses into small fragments of the fakoigla, through the hollow central opening of which they are aspirated due to the suction action of the pump. The success of the operation depends largely on the balance between the processes of cataract mass aspiration and irrigation. The balance between the inflow and outflow of fluid ensures the maintenance of spaces in the anterior segment of the eye, since the whole process takes place in a closed system. It should be noted that the invention of CDKelman was not immediately accepted into clinical practice. At the time of the invention of phacoemulsification, the most popular cataract removal technique was cryoextraction (lens freezing and removal with a capsule through a 12 mm incision), intraocular lenses were rarely implanted, and aphakia correction was essentially the prescription of spectacle lenses. Phacoemulsification changed all the canons. The widespread introduction of microsurgery (i.e., operations under an increase in the operating microscope) and, most importantly, the introduction of small incisions began.
So, the stages of the operation are as follows:
- Access to the lens is performed through the limb zone (where the transparent part of the cornea becomes opaque), two or three small punctures are made — one or two 20G each and one main a little more — 1.8-2.0 mm (depending on the technique that prefers a surgeon). In this zone, the punctures grow very well, it is convenient to work and it is difficult to find them in a couple of weeks even under a microscope.
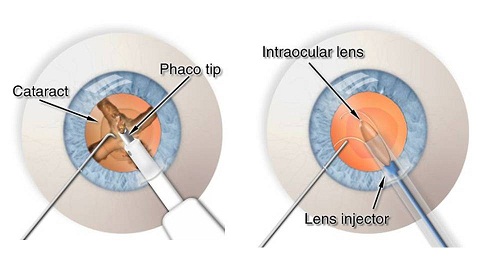
- Through the first main puncture, an ultrasonic tip is inserted with a silicone “sleeve” on the outside. Through the second entrance of smaller diameter — a manipulation chopper (a “poker” for dividing the lens into fragments). The cap is supplied with a sterile special liquid balanced by salt and alkaline composition, and fragments of the crystalline lens, which are emulsified with ultrasound, are sucked into the needle itself. Hence the name — phacoemulsification — “facos” — the crystalline lens, emulsium — a dispersion medium consisting of microscopic droplets dissolved in a liquid.
- Ultrasonic «needle», which performs reciprocating and oscillating movements with a frequency of 28-40 kHz, the lens tissue is crushed to a slurry, which draws in the aspirator. Choppers perform fractures, adhere to fragments in order to feed them into the ultrasonic zone. If the lens is soft, then its removal occurs mainly due to aspiration. If the crystalline lens is dense, then the solid fragments are first mechanically broken up into large fragments, and these in turn are split up by ultrasound to micro-dimensions, in order to remove the aspirator through the tube.
- When the entire dense part of the core is removed, the aspiration and irrigation system removes the softer outer layers and polishes the capsule bag, which must be cleaned as thoroughly as possible. She, like a hammock, is connected by bundles with the rest of the eye.
- An artificial crystalline lens is injected (introduced by a special injector) into this capsule bag. In the tightly folded state of a tight roll. The “legs” are flexed to form something like a single base, assembled when they are closed. Such a designer is a fee for a thin incision. The surgeon fills it in a capsular bag, straightening each leg. The legs (supporting elements) are oval, square or triangular — depending on what kind of lens architecture is used. The main task is to make sure that cells from below that remain from the own lens do not germinate under the lens — they can cause a so-called “secondary cataract,” that is, another clouding. Therefore, various tricks are used — for example, a right angle on the edge of the implant does not allow the cells to “roll over” through it, and the water-repellent coating does not allow to cling to the implant. Artificial lenses made of material with “shape memory” unfold and take on their natural configuration. It is very important how many legs the lens has and how elastic they are to keep the lens in the correct position in the future with not always strong lens bundles.
During the operation there are no important and unimportant stages — so until the last moment until the blepharostat has been removed (the spring that keeps the eyelids open during the operation), the surgeon must keep the situation under control. There is a microsurgeon sitting on top of the patient, while the operation involves two hands, whose actions are controlled visually through the eyepieces of the microscope with a 5-25 fold magnification, two legs — one leg (and heel and sock) are needed to control the microscope (magnification, micro-flooding, regulation illumination of the operative field, centering the image along the XY axis), the second leg controls the operating parameters of the machine (aspiration level, irrigation height, ultrasound amount, vacuum level, change of operating modes). Since the surgeon looks into the eyepieces of a microscope during an operation, he does not have the opportunity to see the instrument panel and performance indicators,
The surgeon does not have assistants and assistants who can intervene in the main process, sitting next to him can only moisten the cornea, dry something or hold it. Therefore, even the only broken toe of the surgeon will become an obstacle to the standard course of the operation — pedals are needed. Everything should be in perfect working condition — both hands with all fingers, both legs, excellent binocular vision (no matter with correction or with glasses — the microscope adjusts to the surgeon's optics) and good hearing (information in the interface is also transmitted via the audio channel). In addition, sufficient exposure, reaction rate and sufficient years of experience.

Complications associated with this type of operation can be many — caused by inadequate technique of operation, non-compliance with device operation modes or incorrect settings, increased intraocular pressure, damage to intraocular structures, inadequate anesthesia, etc. — this is during surgery.
All complications associated with the abdominal type of surgery are possible — inflammation, impaired eye tone (hypertension or hypotension), allergic reactions, vascular reactions, etc. — in the early postoperative period. In the late postoperative period, problems of astigmatism, secondary cataract, and IOL dislocation are possible. It should be noted that we are able to manage the predominant majority of these states.
In order to minimize the risk of these complications, everything must be according to technology.
The result of the operation depends not only on the experience of the surgeon, calculated in years of work, but also on the surgeon’s ability to cope with all non-standard and complex situations that arise during the surgery. Experience decides. Since I have to perform a lot of reconstructive operations after unsuccessful operations performed in other clinics — extract fragments of “fallen” own crystalline lenses from the fundus, displaced artificial lenses, hem, suture, apply the retina — do not hesitate to ask your surgeon how confident he is successful completion of the operation and what risks he sees. The experience and skills of the surgeons are different — perhaps there is another one who will cope with the task better.
How much to wait?
The phrase: “A cataract is not a fruit, it’s not necessary to wait for its ripening” has already become commonplace.
But still the doctors in the clinics say to the patients that the cataract is either initial or still “immature” and is sent to wait. No need to wait! There is nothing to wait for — it will not be better, a cataract, increasing, will force the surgeon to use more ultrasound energy, which in turn will adversely affect the structures surrounding the lens — the cornea, choroid, «maturation» will weaken the ligamentous apparatus, cause changes in the lens capsule, increase the intraocular pressure. Indications for cataract surgery — reduced vision caused by its presence. And visual acuity here is no longer important. There are, for example, types of posterior encapsular cataracts, when it is necessary to operate with vision 1.0, as the quality of vision suffers.
Sometimes, if you wait an extra couple of months, phacoemulsification will have to be abandoned in favor of extracapsular cataract extraction (EEC). Another wait — and the capsule will also become unusable, and only intracapsular extraction (IEK) will turn out. Naturally, the older the operation is historically, the more difficult and longer the healing process is, which leads to secondary complications. With these «manual» types of operations, a frequent problem is surgically induced astigmatism. The smaller the incision and the more precisely it is located — the better the patient will see after the operation. The larger the incision, the worse the refractive picture will be after rehabilitation. By the way, a little more than 15 years ago I was engaged in the technology of cataract removal through a 1 mm incision, using a bimanual technique and implanting an interesting model of a ThinOptix lens through the same 1 mm.
A decade ago, the technological idea was to destroy the lens through a hole of less than 1 mm formed in the anterior capsule of the lens by rotating microlopes. But while the removal through 1.7-1.8 mm is minimal and optimal.
How much to recover?
This is a very individual process. If the patient arrived at an early stage, then the next day he does not have any restrictions. And if he came in with a cataract that has been growing for decades, then recovery will take longer. In any case, during the month you will need to skip drops.
Here in the last post about the lens there are more details.